How AI in Biotechnology Is Reshaping Drug Discovery and Beyond
The global market for AI in biotechnology reached approximately $3.89 billion in 2025 and is projected to grow at a compound annual growth rate of over 19% through the next decade. That figure alone signals a structural shift, not a passing trend. Biotechnology companies that once spent a decade and billions of dollars bringing a single drug to market are now compressing those timelines by years and cutting costs by hundreds of millions through the strategic application of artificial intelligence. The question facing executives, researchers, and investors in this space is no longer whether AI in biotechnology will reshape the industry, but whether their organization will be among those leading the transformation or scrambling to catch up.
This is not a story about incremental efficiency. AI is fundamentally altering how molecules are designed, how clinical trials are structured, how genomic data is interpreted, and how proteins are engineered for therapeutic and industrial applications. The convergence of exponentially growing biological datasets, advanced computing infrastructure, and increasingly sophisticated machine learning models has created conditions where AI can solve problems that were previously intractable. Companies like Insilico Medicine, Recursion Pharmaceuticals, and Exscientia have demonstrated that AI drug discovery can move from target identification to clinical candidate in months rather than years. This blog will explore the specific technologies driving this revolution, the quantified business impact they deliver, a practical roadmap for implementation, and the honest challenges that companies must navigate to succeed.
The Biotechnology Industry at a Crossroads
The biotechnology sector is one of the most capital intensive industries in the world. Bringing a new drug from initial discovery through regulatory approval has traditionally cost an average of $2.6 billion and consumed between 10 and 15 years. These figures represent not just financial expenditure but also enormous opportunity costs, as resources remain tied up in programs that fail at alarming rates. Historically, roughly 90% of drug candidates that enter clinical trials never receive regulatory approval, making pharmaceutical research and development one of the riskiest investment categories in any sector.
Beyond drug development, the broader biotechnology landscape faces its own set of pressures. Agricultural biotechnology companies are contending with climate change, soil degradation, and the need to feed a global population approaching 10 billion. Industrial biotechnology firms are working to replace petroleum based chemicals with sustainable bio based alternatives, a transition that requires massive optimization of enzymatic processes and fermentation pathways. Across all of these domains, the common thread is data: biological systems generate staggering volumes of information, from genomic sequences and proteomic profiles to metabolomic readouts and phenotypic imaging, and the human capacity to analyze this data has been overwhelmed for years.
The competitive dynamics of the industry have also shifted. Venture capital funding for biotechnology startups has become more selective, with investors demanding clearer paths to profitability and shorter timelines to value creation. Large pharmaceutical companies are under pressure from patent cliffs, with blockbuster drugs losing exclusivity and generic competition eroding revenue. The cost of failure has never been higher, and the margin for error has never been thinner. In this environment, companies that can identify promising drug targets faster, design better molecules, optimize clinical trials, and reduce late stage attrition hold a decisive competitive advantage.
Regulatory complexity adds another layer of difficulty. Agencies like the FDA and EMA are evolving their frameworks to accommodate novel modalities such as gene therapies, cell therapies, and RNA based medicines, but the regulatory pathway for these innovations remains uncertain and expensive to navigate. Meanwhile, data privacy regulations and the ethical implications of genomic data use create compliance burdens that slow the pace of research. The industry needs a technology paradigm that can process complexity at scale, reduce uncertainty, and accelerate decision making across every stage of the value chain.
How AI in Biotechnology Is Transforming the Industry
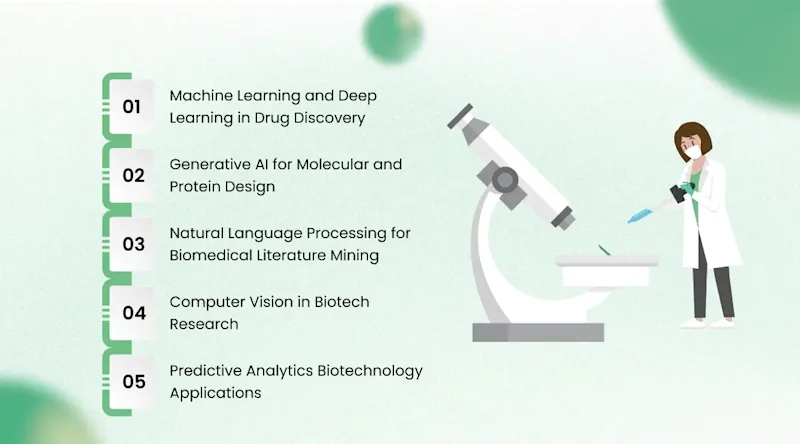
The application of artificial intelligence in biotechnology is not a single technology story. It is an ecosystem of complementary capabilities, each addressing a different bottleneck in the research and development process. Understanding which AI technologies map to which problems is essential for any organization evaluating where to invest.
Machine Learning and Deep Learning in Drug Discovery
AI drug discovery represents the most mature and heavily invested application area. Machine learning models, particularly deep neural networks, are being used to screen virtual libraries of billions of compounds in hours, a process that would take decades using traditional high throughput screening methods. These models learn patterns from known drug target interactions and predict which novel compounds are most likely to bind to a specific protein target with therapeutic relevance. Insilico Medicine demonstrated this capability when it identified a novel target for idiopathic pulmonary fibrosis and advanced a drug candidate to preclinical trials in just 18 months at a fraction of traditional costs. Exscientia, in partnership with Sumitomo Dainippon Pharma, developed a novel molecule for obsessive compulsive disorder in under 12 months, making it the first AI designed molecule to enter human clinical trials.
Deep learning is also transforming toxicity prediction. Bristol Myers Squibb deployed a machine learning program that increased the accuracy of CYP450 inhibition predictions to 95%, representing a sixfold reduction in failure rate compared to conventional screening methods. This capability is critical because late stage safety failures are among the most expensive events in pharmaceutical development, often destroying hundreds of millions of dollars in invested capital. KriraAI works with biotechnology firms to build and deploy these predictive toxicology models, integrating them into existing research workflows so that safety signals are detected at the earliest possible stage.
Generative AI for Molecular and Protein Design
Generative AI has emerged as one of the most transformative technologies in modern biotechnology. Rather than simply screening existing compound libraries, generative models can design entirely new molecules optimized for specific properties such as binding affinity, selectivity, solubility, and metabolic stability. This approach fundamentally expands the chemical space that researchers can explore, moving beyond the limitations of known compounds to create novel therapeutic candidates that would never be discovered through traditional methods.
In AI protein engineering, generative models are now capable of designing novel protein sequences that fold into defined three dimensional structures with predetermined functional properties. The 2024 Nobel Prize in Chemistry recognized work on AI based protein structure prediction and design, underscoring the scientific significance of this capability. Platforms like Cradle Bio use machine learning to design amino acid sequence variants with desired properties such as thermal stability and binding affinity, accelerating the optimization of biologics and industrial enzymes. KriraAI integrates generative AI capabilities into its solutions for biotechnology clients, enabling research teams to move from target identification to optimized lead compounds with significantly fewer experimental cycles.
Natural Language Processing for Biomedical Literature Mining
The volume of published biomedical research doubles approximately every three to four years. No human researcher can keep pace with this flood of information, yet buried within millions of papers are insights about novel drug targets, biomarker associations, and mechanism of action hypotheses that could reshape development programs. Natural language processing models trained on biomedical corpora can extract structured knowledge from unstructured text, identifying relationships between genes, proteins, diseases, and compounds that would take human researchers months to uncover manually. These tools are being used to generate novel hypotheses, validate existing research directions, and identify potential drug repurposing opportunities.
Computer Vision in Biotech Research
Computer vision powered by deep learning is revolutionizing how biological images are analyzed. In high content screening, AI algorithms can analyze thousands of microscopy images per hour, detecting subtle phenotypic changes in cells that human observers would miss. Recursion Pharmaceuticals built its entire platform around this concept, using automated high throughput imaging combined with deep learning to identify phenotypic changes that indicate therapeutic potential. This approach enables rapid screening and repurposing of existing molecules while also discovering novel therapeutics. Computer vision is equally transformative in pathology, where AI models can analyze tissue samples with diagnostic accuracy that matches or exceeds trained pathologists, and in agricultural biotechnology, where drone mounted cameras paired with AI identify crop diseases, nutrient deficiencies, and pest infestations at scale.
Predictive Analytics Biotechnology Applications
Predictive analytics biotechnology applications extend across the entire value chain. In clinical trial design, predictive models optimize patient recruitment by identifying individuals most likely to respond to treatment based on genomic and phenotypic profiles. This reduces trial duration, lowers costs, and improves the probability of demonstrating statistical significance. In manufacturing, predictive analytics monitor bioreactor conditions in real time, adjusting parameters to maximize yield and maintain product quality. In supply chain management, these models forecast demand for biological reagents and raw materials, reducing waste and preventing costly production delays.
Quantified Business Impact of AI Adoption in Biotech
The financial impact of AI in biotechnology is no longer theoretical. Organizations across the industry are reporting measurable improvements in speed, cost, and success rates that directly affect their competitive position and return on invested capital.
AI implementation in preclinical research has been shown to deliver cost reductions of 30% to 70%, primarily through virtual compound screening, predictive modeling, and optimized experimental design. When applied across the pharmaceutical industry at scale, these efficiencies could save between $75 billion and $125 billion annually by 2030. For individual companies, the impact on a per program basis is equally significant. Traditional preclinical programs that cost tens of millions of dollars can be executed for a fraction of that investment when AI guides the selection of compounds, predicts safety liabilities, and optimizes formulation strategies before expensive laboratory work begins.
The impact on clinical development timelines is particularly striking. AI assisted drug development projects have demonstrated timeline reductions of 40% to 60% compared to conventional approaches. In the most accelerated examples, AI has compressed the discovery to preclinical candidate timeline from four to six years down to 12 to 18 months. This acceleration translates directly into earlier revenue generation for successful drugs, extended effective patent life, and faster access to life saving therapies for patients.
Success rates in clinical trials are another area where AI is delivering measurable gains. AI discovered drugs have achieved success rates of 80% to 90% in Phase I clinical trials, compared to historical averages of 40% to 65% for traditionally discovered compounds. This improvement in attrition rates is transformative for portfolio economics, as each late stage failure avoided represents hundreds of millions of dollars in preserved capital that can be redirected to more promising programs.
In protein engineering and biologics development, AI driven design platforms have reduced optimization cycles from months to weeks. Traditional directed evolution experiments require multiple rounds of mutation, expression, screening, and selection, each taking weeks to months. AI models can predict which mutations will improve desired properties before any laboratory work begins, reducing the number of experimental rounds required by 50% to 80%. For companies developing biosimilars, therapeutic antibodies, or industrial enzymes, this acceleration translates into faster time to market and reduced development costs. KriraAI has helped biotechnology clients implement these AI driven optimization workflows, delivering measurable reductions in development cycle time while maintaining the rigorous quality standards required for regulatory submissions.
In genomics, machine learning in biotech has reduced the cost and time required for variant interpretation. What once required expert geneticists spending days analyzing a single genome can now be accomplished in minutes with AI assistance, enabling clinical genomics programs to scale from hundreds to hundreds of thousands of patients without proportional increases in staffing costs.
A Practical Roadmap for AI Implementation in Biotechnology
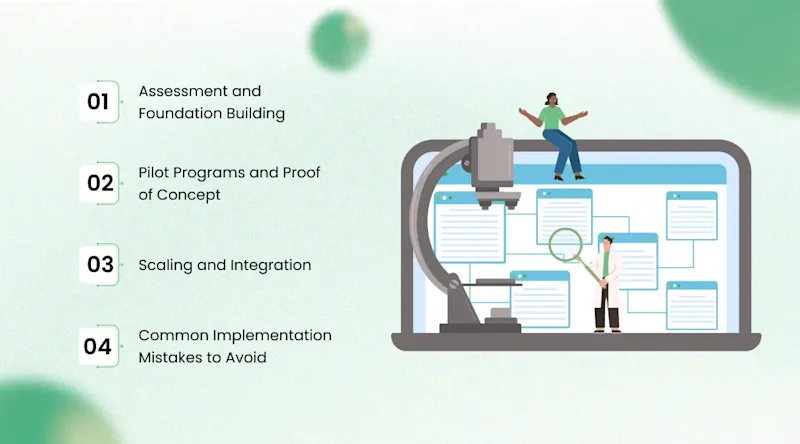
Implementing AI in a biotechnology organization is not as simple as purchasing software. It requires a structured approach that accounts for data readiness, organizational capabilities, regulatory requirements, and change management. The following roadmap reflects the practical steps that successful adopters have followed.
Phase 1: Assessment and Foundation Building
The first phase involves a thorough audit of the organization's data assets, computational infrastructure, and talent capabilities. This assessment should answer several critical questions.
What biological and experimental data does the organization currently generate, and in what formats and systems is it stored?
How clean, standardized, and accessible is this data, and what gaps exist in data quality or coverage?
What computational infrastructure is available, including cloud computing resources, GPU capacity, and data storage?
What AI and data science talent exists within the organization, and what skills need to be recruited or developed?
Which business problems represent the highest value opportunities for AI, considering both potential impact and feasibility?
This phase typically takes two to three months and should produce a prioritized list of AI use cases ranked by expected value and implementation complexity. Organizations that skip this phase often waste significant resources building solutions to the wrong problems or discovering mid project that their data is insufficient to train effective models.
Phase 2: Pilot Programs and Proof of Concept
The second phase focuses on executing one to three carefully selected pilot programs that can demonstrate measurable value within six to twelve months. The best pilot programs share several characteristics: they address a genuine business need, they have access to sufficient high quality data, they have an engaged internal champion, and their success can be measured with clear metrics.
Common pilot areas in biotechnology include virtual screening of compound libraries for a specific target, predictive modeling for ADMET properties (absorption, distribution, metabolism, excretion, and toxicity), automated analysis of microscopy or pathology images, and NLP based mining of scientific literature for target validation. The pilot phase should include rigorous validation against experimental results and comparison with existing methods to quantify improvement.
Phase 3: Scaling and Integration
Successful pilots should be scaled into production systems integrated with existing research workflows. This phase involves engineering work to connect AI models with laboratory information management systems, electronic lab notebooks, and data pipelines. It also requires establishing model governance frameworks including version control, performance monitoring, retraining schedules, and documentation for regulatory submissions. Organizations at this stage often benefit from partnering with experienced AI implementation firms like KriraAI, which specializes in building production grade AI systems that integrate seamlessly with established biotechnology workflows.
Common Implementation Mistakes to Avoid
Several recurring mistakes derail AI initiatives in biotechnology, and awareness of these pitfalls can save organizations significant time and resources.
Starting with the technology rather than the problem is the most common failure mode, resulting in impressive demonstrations that deliver no business value.
Underinvesting in data engineering leaves organizations with AI models built on unreliable foundations, producing predictions that cannot be trusted for decision making.
Failing to involve domain scientists in model development produces technically sophisticated but scientifically naive solutions that miss critical biological context.
Neglecting change management creates resistance from researchers who view AI as a threat rather than a tool, leading to low adoption even when the technology works.
Ignoring regulatory requirements from the outset creates compliance risks that can delay or prevent the use of AI derived insights in regulatory submissions.
Challenges and Limitations of AI in Biotechnology
Honest assessment of the challenges facing AI adoption in biotechnology is essential for realistic planning and expectation management. The technology is powerful but far from a universal solution, and organizations that underestimate these challenges risk costly failures.
Data quality remains the single most significant barrier to effective AI deployment. Biological data is inherently noisy, heterogeneous, and often poorly standardized across laboratories and organizations. Historical experimental data may be stored in incompatible formats, lack critical metadata, or contain systematic biases that propagate through AI models. Building the data infrastructure required for reliable AI applications often takes longer and costs more than the AI development itself. Organizations must invest in data governance, standardization, and curation before expecting meaningful returns from AI.
The talent gap in biotechnology is acute. Effective AI implementation requires individuals who understand both the computational techniques and the biological domain, a combination of skills that is exceedingly rare. Pure data scientists often lack the biological intuition needed to build models that capture meaningful biology, while bench scientists may lack the computational skills to evaluate or work with AI tools effectively. Building interdisciplinary teams that bridge this gap requires deliberate organizational design, competitive compensation, and sustained investment in training.
Regulatory uncertainty creates additional friction. While agencies like the FDA have signaled acceptance of AI in drug development, with over 500 drug applications containing AI components submitted between 2016 and 2023, the regulatory framework for validating and documenting AI derived decisions is still evolving. The EU AI Act introduces new compliance obligations that will affect biotechnology companies operating in or serving European markets. Organizations must design their AI systems with regulatory documentation in mind from the beginning, not as an afterthought.
Integration complexity should not be underestimated. Biotechnology organizations typically operate with complex technology ecosystems including laboratory instruments, data management systems, quality management systems, and regulatory submission platforms. Connecting AI models to these systems in a reliable, validated, and maintainable way requires substantial engineering effort and ongoing operational support. The total cost of ownership for AI systems extends well beyond the initial development, encompassing maintenance, retraining, monitoring, and integration management.
The Future of AI in Biotechnology Over the Next Five Years
The trajectory of AI in biotechnology over the next three to five years points toward a fundamental restructuring of how biological research and development is conducted. Several trends are converging to accelerate this transformation beyond what most industry observers currently anticipate.
Autonomous laboratories represent one of the most significant near term developments. These facilities combine robotic automation with AI decision making to design experiments, execute them physically, analyze results, and plan the next experimental cycle with minimal human intervention. Early versions of these systems are already operational at companies like Recursion and Emerald Cloud Lab, and the technology will mature rapidly as robotics costs decline and AI planning algorithms improve. Within five years, autonomous lab systems will be accessible to mid sized biotechnology companies, not just well funded technology pioneers.
The convergence of AI with CRISPR based gene editing will unlock capabilities that neither technology could achieve alone. AI models will predict the outcomes of specific genetic edits with high accuracy, enabling precision engineering of cell lines, model organisms, and eventually therapeutic gene therapies with unprecedented specificity and safety. This combination will accelerate the development of cell and gene therapies, which represent the fastest growing segment of the biopharmaceutical pipeline.
Foundation models trained on biological data will become as transformative for biotechnology as large language models have been for text based applications. These models, trained on massive datasets encompassing genomic sequences, protein structures, metabolic pathways, and clinical outcomes, will provide general purpose biological intelligence that can be fine tuned for specific applications. Early examples like AlphaFold have demonstrated the potential of this approach for protein structure prediction, but the next generation of biological foundation models will extend to predicting protein function, designing metabolic pathways, and simulating cellular responses to perturbation.
Companies that fail to build AI capabilities within the next two to three years will find themselves at a structural disadvantage. The gap between AI enabled organizations and traditional organizations will widen as AI driven companies accumulate proprietary data assets, refine their models through iterative learning, and compress their development timelines to outpace competitors. This is not a technology arms race that can be won by late adoption; the compounding nature of AI improvement means that early movers build advantages that become increasingly difficult to replicate.
Conclusion
Three key insights emerge from this analysis of AI in biotechnology. First, the technology has matured beyond proof of concept into a practical tool delivering measurable cost reductions, timeline compression, and improved success rates across drug discovery, genomics, protein engineering, and clinical development. Second, successful implementation requires a disciplined approach that prioritizes data readiness, domain expertise, and organizational change management over technology acquisition. Third, the competitive window for adopting AI is narrowing, as early movers accumulate compounding advantages in data, models, and operational efficiency that will be increasingly difficult for late adopters to overcome.
For biotechnology organizations ready to act on these insights, the path forward requires a partner that understands both the science and the technology. KriraAI builds practical AI solutions for biotechnology enterprises, combining deep domain expertise with production grade engineering to deliver systems that integrate with existing research workflows and meet the rigorous validation standards required for regulatory submissions. Whether the objective is accelerating drug discovery pipelines, optimizing protein engineering workflows, or building predictive analytics capabilities for clinical development, KriraAI's approach focuses on measurable outcomes rather than abstract innovation.
The biotechnology companies that will lead the next decade are those making strategic AI investments today. If your organization is evaluating how AI can strengthen its research and development capabilities, exploring KriraAI's solutions is a practical next step toward building the data driven competitive advantage that this industry increasingly demands.
FAQs
Artificial intelligence is used across nearly every stage of the biotechnology value chain, from early research through manufacturing and commercialization. In drug discovery, AI models screen billions of virtual compounds to identify those most likely to bind therapeutic targets, reducing the time and cost of identifying lead candidates by orders of magnitude compared to traditional high throughput screening. In genomics, machine learning algorithms analyze DNA sequences to identify disease associated variants, predict gene function, and guide the development of precision therapies tailored to individual genetic profiles. In protein engineering, generative AI designs novel amino acid sequences with specified structural and functional properties, accelerating the development of therapeutic antibodies, enzymes, and biosimilars. Beyond pharmaceuticals, AI is used in agricultural biotechnology for crop trait optimization, in industrial biotechnology for fermentation process optimization, and in clinical settings for diagnostic image analysis and biomarker discovery.
The cost savings from AI in drug discovery are substantial and well documented across multiple stages of the development process. AI implementation in preclinical research has been shown to deliver cost reductions ranging from 30% to 70%, primarily through virtual compound screening that eliminates the need for expensive physical assays, predictive toxicology models that catch safety issues before costly animal studies, and optimized experimental designs that reduce the number of iterations required to identify promising candidates. Across the pharmaceutical industry at scale, these efficiencies could collectively save between $75 billion and $125 billion annually by 2030. For individual drug programs, AI can reduce the total cost of bringing a compound from discovery to clinical candidate by 40% or more, translating to savings of hundreds of millions of dollars per successful program. Cloud computing solutions further amplify these savings by reducing infrastructure costs by 40% to 60%, while open source AI tools can lower licensing expenses by 30% to 50%.
Implementing AI in a biotechnology company is a phased process that typically spans 12 to 24 months from initial assessment to production deployment, depending on the organization's data readiness, infrastructure maturity, and the complexity of the chosen use cases. The initial assessment and foundation building phase, which includes data auditing, infrastructure evaluation, and use case prioritization, generally requires two to three months. Pilot programs and proof of concept development typically take an additional six to twelve months, including model training, validation against experimental results, and stakeholder review. Scaling successful pilots into production systems integrated with existing workflows adds another three to six months, encompassing engineering work, regulatory documentation, and change management activities. Organizations with mature data infrastructure and experienced teams can move faster, while those starting from scratch should plan for the longer end of this range and invest heavily in data engineering before attempting to build sophisticated models.
Machine learning plays an increasingly central role in genomics across the biotechnology industry, serving as the primary analytical engine for extracting meaningful insights from the vast quantities of sequence data generated by modern high throughput platforms. In clinical genomics, ML algorithms classify genetic variants as pathogenic or benign, a task that traditionally required expert human geneticists spending days per genome and can now be accomplished in minutes with AI assistance. In functional genomics, deep learning models predict how specific genetic variations affect RNA splicing, protein expression, and cellular phenotype, enabling researchers to prioritize variants for experimental validation and therapeutic targeting. In population genomics, machine learning identifies patterns across large cohorts that reveal novel gene disease associations, pharmacogenomic relationships, and evolutionary signatures. These capabilities are foundational to the development of precision medicine, where treatments are tailored to individual patients based on their unique genetic profiles, and they underpin the growing field of predictive diagnostics that aims to identify disease risk before symptoms appear.
AI will not replace scientists in biotechnology research, but it will fundamentally change the nature of their work and dramatically amplify their capabilities. The most accurate way to understand AI's role is as a force multiplier that handles computationally intensive tasks such as screening millions of compounds, analyzing thousands of images, or processing vast genomic datasets, freeing scientists to focus on the creative, interpretive, and strategic aspects of research that require human judgment, biological intuition, and interdisciplinary thinking. Scientists who learn to work effectively with AI tools will be far more productive than those who do not, capable of testing more hypotheses, exploring larger experimental spaces, and making more informed decisions in less time. The demand for scientists with hybrid skills combining domain expertise with computational literacy is growing rapidly, and organizations that invest in developing these capabilities within their research teams gain a significant competitive advantage. The future of biotechnology belongs to teams where human creativity and AI analytical power work in concert, not to either one operating in isolation.
Founder & CEO
Divyang Mandani is the CEO of KriraAI, driving innovative AI and IT solutions with a focus on transformative technology, ethical AI, and impactful digital strategies for businesses worldwide.
